Harald Sontheimer awarded Commonwealth of Virginia Alzheimer’s Research Grant
August 3, 2017
The Alzheimer's and Related Diseases Research Award Fund (ARDRAF) was established by the Virginia General Assembly in 1982 to stimulate innovative investigations into Alzheimer's disease and related disorders along a variety of avenues, such as the causes, epidemiology, diagnosis, and treatment of the disorder; public policy and the financing of care, and the social and psychological impacts of the disease upon the individual, family, and community. The awards this year have been enhanced by a $25,000 donation from Mrs. Russell Sullivan of Fredericksburg, in memory of her husband who died of dementia. The ARDRAF competition is administered by the Virginia Center on Aging in the School of Allied Health Professions at Virginia Commonwealth University.
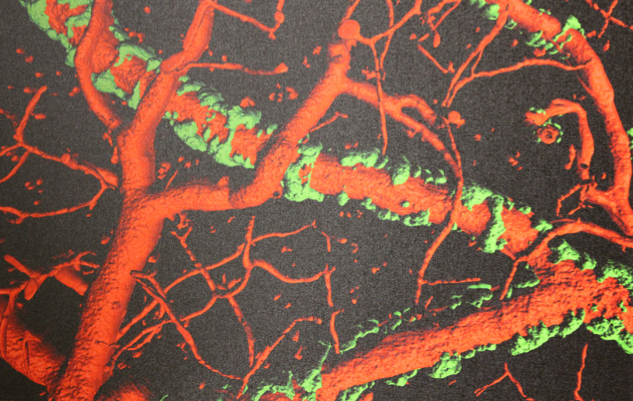
Harald Sontheimer’s research group has studied Amyloid proteins and their impact on Alzheimer disease for sometime. It is commonly assumed that amyloid contributes to functional impairment of neurons, albeit how amyloid is toxic to brain remains unclear. While plaques are found near neurons, they are often close to brain support cells called astrocytes as well as along blood vessels. The astrocytes touch blood vessels throughout the brain and have been shown to support the integrity of the blood brain barrier (BBB) that prevents entry of blood born molecules into the brain. Astrocytes also regulate blood flow by releasing vasoactive molecules. The investigator has demonstrated that vascular amyloid separates the astrocytic attachments on blood vessels called endfeet. By forming a rigid cast around arterioles and penetrating arteries, the amyloid deposits hinder the release of vasoactive molecules and impair the regulation of blood flow. This study will explore whether amyloid deposits also cause local impairment of blood flow and BBB integrity. The over-arching hypothesis is that amyloid impairs astrocyte function and, therefore, vessel health and local regulation of blood flow. Obviously, impairments of blood flow will starve neurons of energy and could hasten their demise, thereby explaining the progressive dementia. These studies may show a completely unexplored pharmacological target.
Sontheimer said if his team’s hunch is true, it could serve as a “paradigm shift” in Alzheimer’s research. Research work will be carried out in labs with Institute for Creative Technology and Applied Science, with Ian Kimbrough, a post-doctorate researcher with the Virginia Tech Carilion Research Institute and instructor with the School of Neuroscience, and Tre Mills III, a doctorate researcher with the Virginia Tech Graduate School’s translational biology, medicine, and health program, assisting Sontheimer.
Sontheimer expects a follow-up National Institutes of Health grant to continue funding his research.