Core Faculty: Research Opportunities
Agrawal Laboratory
Proprioceptive control of movement
The Agrawal lab studies how sensory feedback from proprioceptive sensory neurons enables the flexible control of behavior. We focus on the compact nervous system of the fruit fly, Drosophila melanogaster and utilize a variety of techniques, including quantitative behavior, optogenetics, in vivo electrophysiology and 2-photon imaging, EM-based circuit reconstruction, comparative morphology, and computational modeling. We are also interested in understanding how motor context or limb morphology shape proprioceptive circuits, especially in non-Drosophila insects.
Agrawal Laboratory

Buczynski Laboratory
Identifying druggable targets for treating addiction
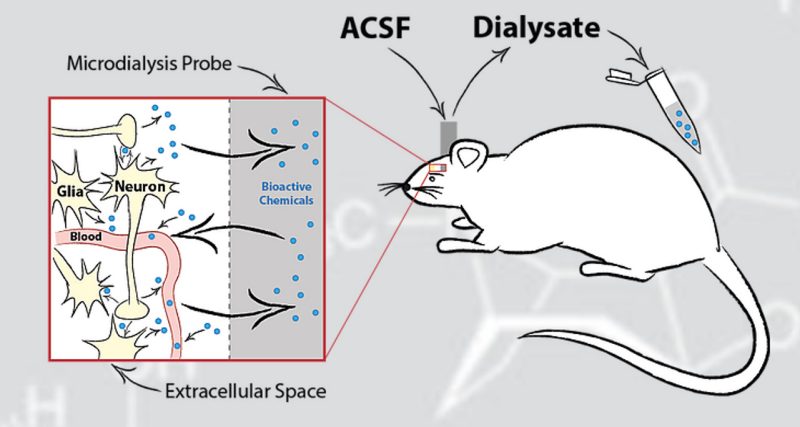
Our laboratories fundamental research objective is to identify the novel molecular changes caused by drug exposure and validate their functional role in neurological disorders. The research integrates advanced mass spectrometry platforms with a wide range of techniques including chemical biology, molecular biology, in vivo microdialysis, and behavioral pharmacology.
Schematic of in vivo microdialysis probe setup, neurochemical diffusion, and sample collection
English Laboratory
Neuronal representation of space and time
Our research examines how interactions between excitatory and inhibitory neurons generate organized activity in neural circuits. In particular, we are interested in: (1) how excitatory and inhibitory neurons in the CA1 region of the hippocampus generate representations of space; (2) the synaptic and circuit mechanisms of learning-related sharp-wave ripple oscillations in hippocampus and connected cortical regions, especially the retrosplenial cortex. To address these questions we combine electrophysiology, optogenetics and 1- and 2-photon imaging, in awake behaving mice.
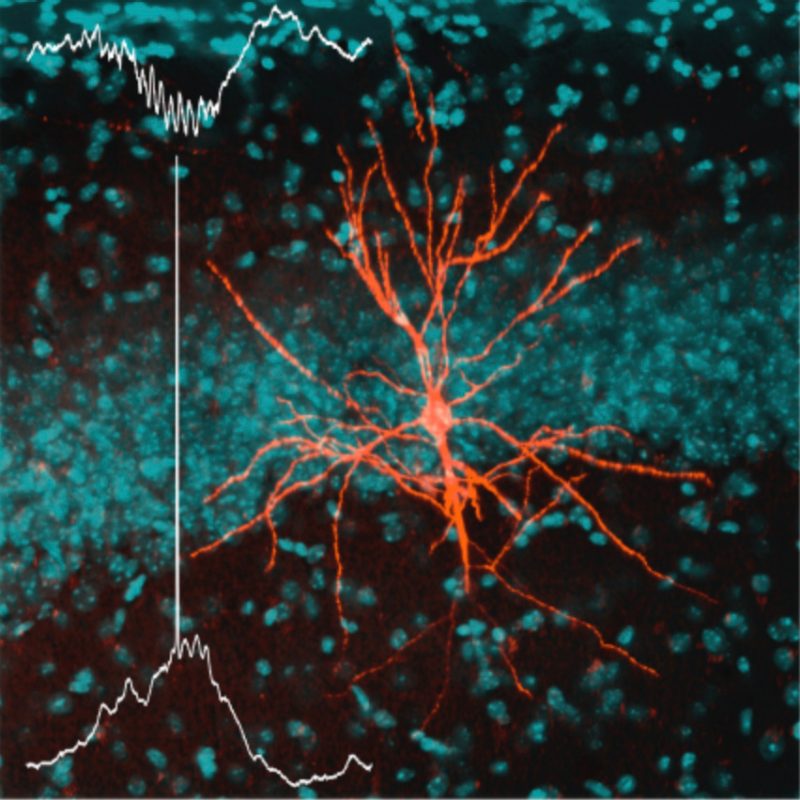
Gregus Laboratory
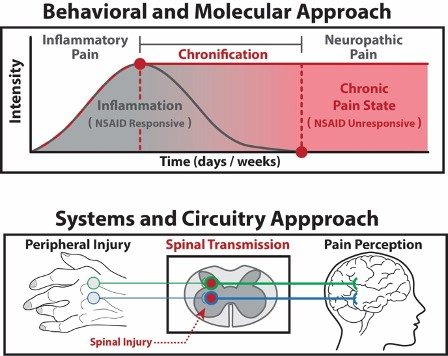
Drug discovery for chronic pain states
Chronic pain affects 30-40% of Americans, most of whom report that their pain is inadequately managed by current treatments. While opioids are effective for moderate to severe pain, they carry risks of addiction. Thus, our goal is to elucidate novel molecular mechanisms driving the transition from acute to chronic pain and to translate druggable targets into potential nonopioid therapeutics via a multi-omic approach using advanced mass spectrometry techniques, cell-based assays, and behavioral pharmacology to support early-stage drug discovery.
Hodes Laboratory
Resilience to stress and mood disorders
The goal of our lab is to identify biological mechanisms that contribute to individual differences in vulnerability and resilience to stress and mood disorders such as anxiety and depression. We are working to develop novel personalized treatments and bioassays for mental illness so these disorders can be medically diagnosed and treated effectively. Our research program examines sex differences in the peripheral and central immune system and how immune mechanisms interact with brain plasticity to drive behavioral differences in response to stress.
Howe Laboratory
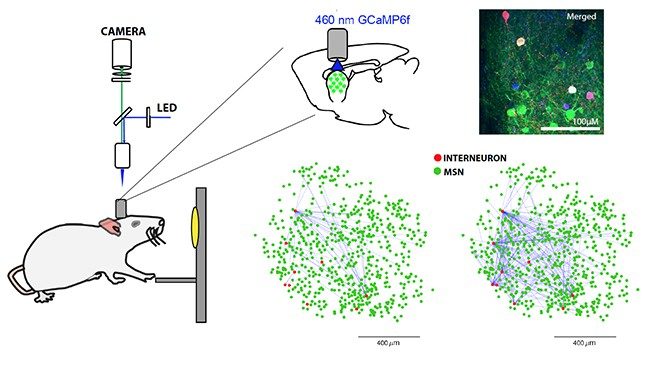
Neural circuits of motivation and attention
Dysregulation of the neurobiological mechanisms that support fundamental functions like motivation and attention may contribute to cognitive and behavioral symptoms of neuropsychiatric and neurodegenerative disorders. Research in the Howe lab takes a systems neuroscience approach to identify the brain-circuits that control these functions, as well as targets within these circuits that can guide the development of new therapeutics.
Jarome Laboratory
Molecular and cellular biology of memory formation
Our research focuses on the neurobiology of learning and memory with an emphasis on understanding the molecular and epigenetic mechanisms involved in posttraumatic stress disorder (PTSD). Additionally, we also investigate the molecular and epigenetic mechanisms involved in neurodevelopmental disorder and obesity. We address these topics using rodent behavioral paradigms in combination with a wide variety of traditional and modern molecular genetic techniques, including CRISPR-dCas9.
Legon Laboratory
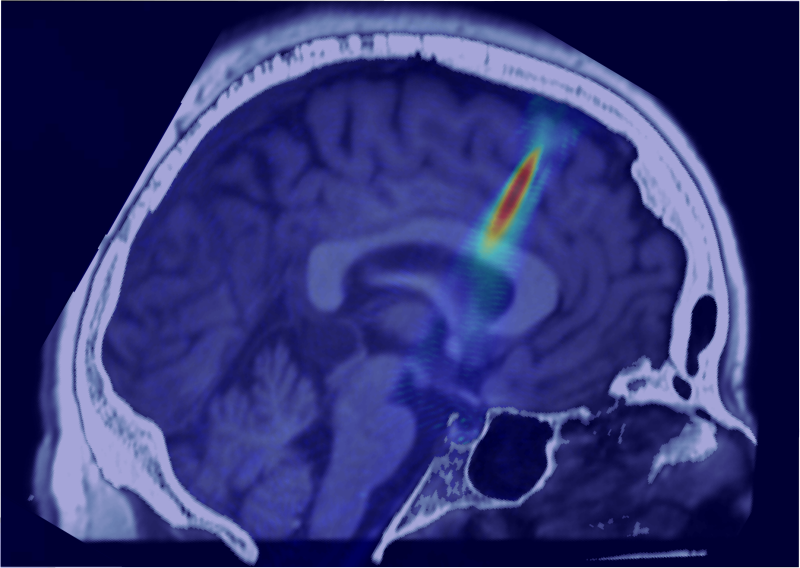
Low-intensity focused ultrasound
The LegonLab studies the use of low-intensity focused ultrasound (LIFU) for human neuromodulation. We use a multi-modal approach including fMRI, EEG and TMS as well as physiological measures of heart-rate, HRV, electrodermal response and continuous blood-pressure combined with behavioral metrics to understand how LIFU affects brain activity and autonomic function. The lab also conducts empirical acoustic testing and acoustic modelling of ultrasound wave propagation.
Ni Laboratory
Neuronal circuitry of thermoregulation
Our research focuses on understanding how sensory systems regulate behavior. Our lab focusses on sensory transduction and is trying to address the following three questions: How do environmental stimuli activate the sensory receptors? How does the information from these receptors be processed in the brain? And how does the brain control behavior and physiology?
Olsen Laboratory
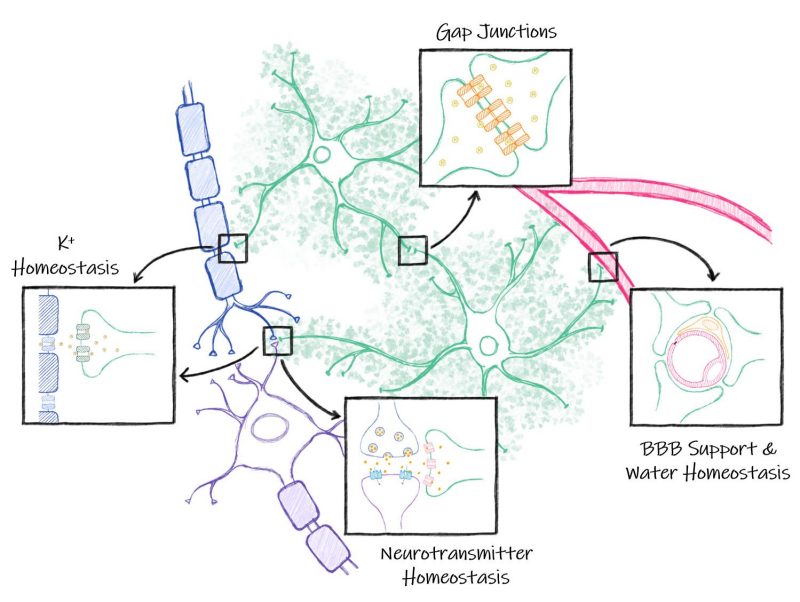
Astrocyte function in health and disease
The main interest of the Olsen lab is to better understand the role of astrocytes in normal and abnormal central nervous system function. Projects in the Olsen lab are aimed at understanding how astrocytes contribute to development and maturation of the central nervous system.
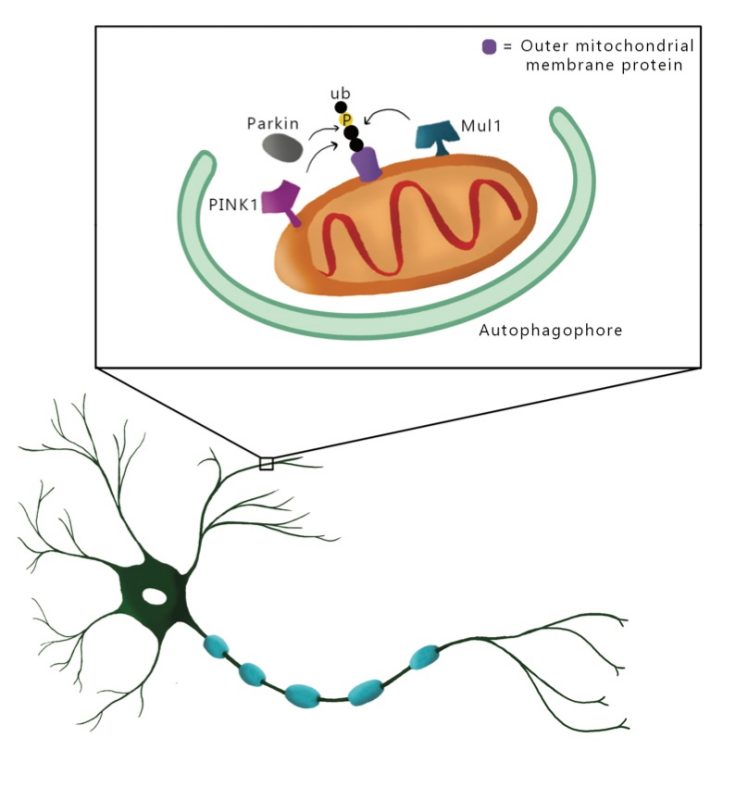
Pickrell Laboratory
Mitochondrial dysfunction in neurodegeneration
The focus of our research is to understand how and why neurons become vulnerable during disease states. My prospective research projects will focus on: 1) to what degree mitochondrial dysfunction is tolerated by different neuron subtypes, 2) mechanisms that contribute to the accumulation of mutated mtDNA in neurons, and 3) to what extent mitochondrial function is affected by aggregates that accumulate in neurodegenerative disease, such as aIpha-synuclein.
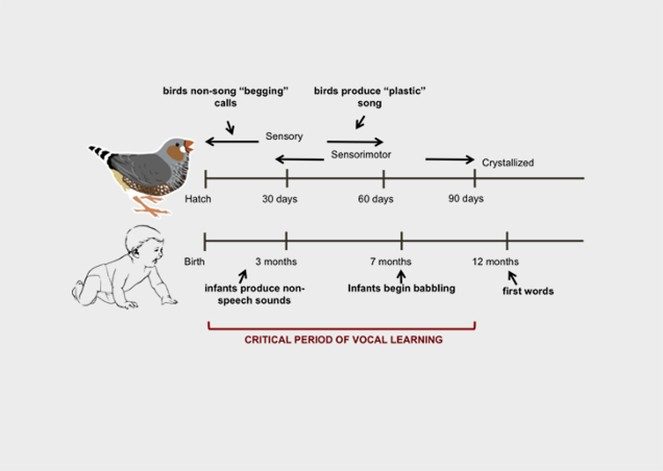
Sewall Laboratory
Environmental effects on brain and behavior
The Sewall lab studies how the environment impacts the brain and behavior of songbirds. Songbirds are highly social and they learn their vocalizations, making them excellent for studying sociality, learning, and communication. Our current work focuses on how urbanization, social dynamics, and heavy metal exposure impact these complex behaviors through effects on brain growth and neuroendocrine mechanisms.
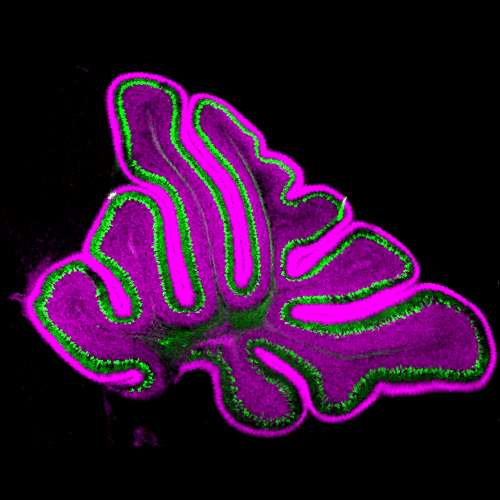
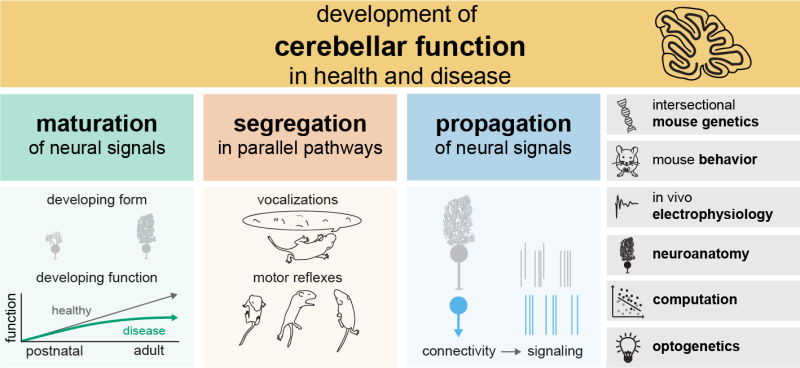
Van der Heijden Laboratory
Development of cerebellar function in health and disease
We study the development of cerebellar function in health and disease. Abnormal cerebellar development is associated with pediatric movement disorders, like ataxia and dystonia, and neurodevelopmental disorders, like autism spectrum disorder. We seek to understand how the healthy cerebellum contributes to the postnatal development of behaviors so that we can predict, prevent, and reverse neurodevelopmental disabilities caused by abnormal cerebellar development.
Vijayan Laboratory
Neural dynamics and neural engineering
Our laboratory investigates the neural dynamics underlying sleep-mediated memory consolidation in health and disease (e.g., Parkinson’s Disease and posttraumatic stress disorder), with additional projects on sensory processing, sleep processes, brain machine interfaces (BMI), and mental imagery. We employ both invasive (intracranial) and non-invasive (EEG) techniques to examine neural activity and signal processing and computational modeling techniques to make sense of the data we collect.
Weston Laboratory
Physiological underpinnings of neurodevelopmental disorders
Physiological underpinnings of neurodevelopmental disorders.
Our research program aims to understand how genetic variants that cause epilepsy, and other neurological diseases, impact synaptic function and intrinsic excitability, and how these neuronal changes lead to aberrant network activity and seizures. Our guiding hypothesis is that changes in synaptic and membrane function are key events in the pathway that leads from a genetic abnormality to disease phenotypes and that identifying these changes and their impact on neural circuit function are essential for a basic understanding of normal and pathological brain function. To achieve this, our lab interrogates functional alterations in genetic mouse models of epilepsy using imaging and electrophysiology techniques, both in vitro and in awake, behaving mice.